
July 1996 Volume 2 Issue 12
ENERGY RECOVERY FROM HOT LIQUIDS
In food processing plants, hot liquids are often sent to drain. In many cases this is just where the liquid should go — but must the heat energy which the liquid contains go down the drain with it? Sometimes not. In this issue of $mart Energy User we explore the possibility of reducing fuel consumption by recovering some of that energy.
DEALING WITH STEAM CONDENSATE
Steam is an extremely convenient heat source. It moves easily from one place to another through pipes, and it carries a great deal of heat energy with it. Most of that heat energy is released as the steam condenses into liquid water — but not all of it. Immediately after condensing, the liquid condensate is still at or near steam temperature and contains potentially useful energy.
In “closed” steam systems the steam does not make direct contact with food but transfers heat to it indirectly via heat exchangers, steam-jacketed cooking utensils, immersed steam coils, etc. As heat is transferred to the food, steam condenses within the closed system and appears at the condensate outlets of steam traps. From there, the condensate is piped back to the boiler where it provides most of the feedwater needed for additional steam generation. Recycling the hot condensate in this manner keeps energy costs down.
In “open” steam systems the steam makes direct contact with the food. In some cases steam is sparged into a vessel containing both water and the food to be cooked (lobster or crab, for example). Alternatively, the food is moved (without water) through a semi-enclosed space containing steam at atmospheric pressure. In both cases steam condenses into hot water, but this condensate is mixed with food juices, salt, and perhaps other contaminants, and is thus unsuitable for direct use as boiler feedwater.
Food-contaminated condensate is often sent directly to drain, taking considerable heat energy with it. This is unfortunate, since there are ways of recovering that heat, and there are good uses for it in many plants.
IS THERE A USE FOR THE RECOVERED HEAT?
For heat recovery to make economic sense there must be a use for that heat which actually reduces the amount of fuel or electricity purchased.
Two uses for reclaimed heat that make sense in many plants are preheating boiler feedwater and preheating wash water. Water from P.E.I. wells is quite cold — usually about 45°F (7°C). For this water to become steam or wash water, it must be heated from this low temperature to a much higher temperature:
· to 212°F (100°C) or higher in the case of steam, and
· to between 115°F (45°C) and 180°F (82°C) in the case of wash water.
Normally, fuel is burned to do this. Sometimes electricity is used to heat wash water.
The amount of energy needed to heat water is proportional to the difference between the initial and final temperature of the water. Let’s assume we are starting with 7°C well water and our goal is to produce 77°C wash water. The cost of doing all that heating with #2 oil at $0.25 per litre and 80 per cent combustion efficiency would be $2.37 for each
1000 litres of water heated. If, however, reclaimed heat could be used to raise the water temperature the first 35°C — from 7°C to 42°C — then only half as much fuel would be needed to heat it the rest of the way, and the cost of heating the wash water would be cut in half.
IS THERE ENOUGH HEAT AVAILABLE?
The economic feasibility of heat recovery also depends on whether there is enough recoverable heat available. When considering the heat recovery potential of an effluent flow, the important factors are the temperature of the effluent relative to the temperature of the liquid to be heated, and the rate of flow of the effluent. A large temperature difference and high effluent flow rate are desireable. An example may help.
Let’s assume that we have an 85°C effluent flowing at a rate of one litre per second for a total duration of 1000 hours per year. Let’s also assume that we would use this effluent to preheat boiler feedwater, and that in transferring heat to the cold well water the effluent temperature would drop to 55°C. The THEOS Thermal Energy Opportunity Software tells us that this represents an energy flow of 126 kW, and that the energy transferred during those 1000 hours would cost $3658 to produce if we did it by burning #2 oil worth $0.25/litre at 80% combustion efficiency.
HEAT RECOVERY TECHNIQUES
Recovering that $3658 worth of heat would require an investment in heat recovery hardware. Heat recovery involves transferring heat energy from a hot liquid to a cooler one while keeping the two liquids separate. Heat exchange is the technical term for this process, and devices called heat exchangers are often employed to accomplish it. One version of the shell-and-tube heat exchanger design is shown below. In our example, the hot effluent would flow through the small-diameter tubing, and the boiler feedwater would flow through the shell or jacket that surrounds the tubing. A device like this keeps the two liquids separate, yet allows heat to flow from the hotter liquid to the cooler one through the tubing walls.
In a variation on the heat-exchanger idea, a substantial length of tubing is immersed in a tank of water. The tank holds the water being preheated — the wash water or boiler feedwater. The hot effluent flows through the submerged tubing on its way to drain. The effluent loses heat to the surrounding water and cools as it passes through the tubing. At the same time the water in the tank gains heat and rises in temperature.
GETTING HELP
If you think there might be some potential for heat recovery in your plant, contact us and we will work with you on an initial evaluation. Call Mike Proud or Ron Estabrooks at 368-5010 (toll free). You might also be interested in obtaining the Windows®-based THEOS software mentioned above, as well as our new booklet Energy-Efficient Process Heat. Both are available at no cost to recipients of this newsletter.
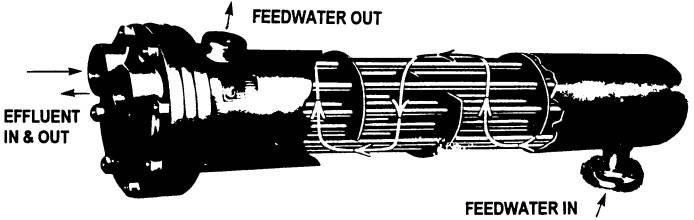
A typical shell-and-tube water-to-water heat exchanger
